Professional graphite material supplier, graphite for EV, grease, furnace and any other industries.
(how is graphite different from other allotropes of carbon)
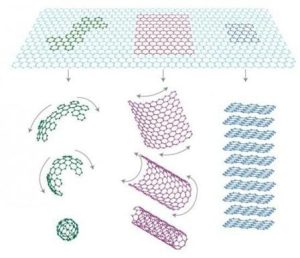
Graphite, also known as carbide or silicified carbon, is a carbon-based mineral that has a very low melting point and high thermal conductivity. It is unique in that it is composed entirely of carbon atoms arranged in a hexagonal lattice structure.
(how is graphite different from other allotropes of carbon)
One of the key differences between graphite and other allotropes of carbon is its electrical conductivity. Graphite is an excellent conductor of electricity due to the presence of free electrons in its crystal lattice structure. This property makes it useful for applications such as electrical batteries and solar panels.
Another important difference between graphite and other allotropes of carbon is its durability. Graphite has a high melting point, which means that it can withstand high temperatures without cracking or breaking. This makes it a popular material for use in temperature-sensitive applications such as electronic components and aerospace components.
Graphite is also lightweight and durable, making it a popular choice for use in various industrial products. For example, it is used in the production of pencils, pens, and brushes due to its ease of writing and drawing. Graphite is also used in the production of diamonds, another famous gemstone.
(how is graphite different from other allotropes of carbon)
Finally, graphitChat hot tags: graphite,graphite powder,nano graphite
(how is graphite different from other allotropes of carbon)