Professional graphite material supplier, graphite for EV, grease, furnace and any other industries.
(How Many Atoms In A Unit Cell Of Graphite)
Graphite is a hard, black, dense rock that forms when coarselyed iron ore is exposed to high temperatures and pressures for a period of time. The term “unit cell” refers to the amount of atoms in one unit cell of graphite, which is the size of a single atom.
(How Many Atoms In A Unit Cell Of Graphite)
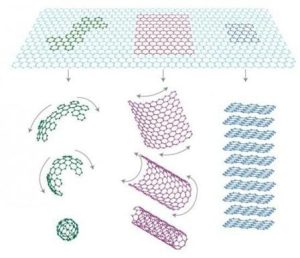
Graphite has a unique composition that makes it suitable for use as an electronic material. It contains several elements such as carbon, silicon, oxygen, and aluminum, making it highly brittle and resistant to decay. These elements combine to form one layer of metal atoms, forming a single layer on top of each other. This layer is called the crystal, and it is characterized by its long chains of bonds.
The atomic number of graphene is a combination of three different elements: iron, silicon, and oxygen. Iron gives it a good resistance to rusting and corrosion, while silicon makes it light and resistive. Oxygen gives it a wide range of colors and a rich chemical compound, making it useful in a variety of applications, including electronics, semiconductors, and even batteries.
One of the main challenges facing scientists in studying graphene is how to control its properties and behavior. Researchers have used various methods to study the structure and behavior of graphene, but there are still many unanswered questions about its behavior under extreme conditions, such as extreme temperatures and pressures. They also need to develop new techniques to make graphene functional and practical, such as using it in solar cells or drug delivery systems.
Despite these challenges, scientists continue to explore the potential uses of graphene in a variety of fields. For example, researchers have used graphene in the development of insulin-like agents that can improve insulin sensitivity and reduce blood sugar levels in patients with diabetes. Additionally, graphene has been used to create artificial skin and other surfaces that are difficult to otherwise produce.
(How Many Atoms In A Unit Cell Of Graphite)
In conclusion, the atomic number of graphene is a powerful tool for scientists to study and understand the properties and behavior of this fascinating material. However, there are still many unanswered questions about its properties and behavior, and researchers must continue to work towards developing new techniques and tools to make graphene functional and practical. hot tags: graphite,graphite powder,nano graphite
(How Many Atoms In A Unit Cell Of Graphite)