Professional graphite material supplier, graphite for EV, grease, furnace and any other industries.
(What Has Stronger Carbon Carbon Bonds Graphite Diamond C60)
Carbon-carbon bonds, or carbon structures, form the backbone of many materials and have been studied extensively for their potential as energy sources. One of the most interesting properties of carbon-carbon bonds is that they can form strong covalent bonds through a process known as intercalation. This allows carbon to be packed closely together in a material, creating a strong network of carbon-carbon bonds.
(What Has Stronger Carbon Carbon Bonds Graphite Diamond C60)
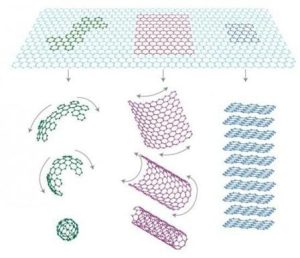
One example of a material that utilizes this type of carbon-carbon bonding is graphite. Graphite is a naturally occurring carbon-rich material that has many applications in science and technology. It is commonly used in pencils, pens, and electronic devices because of its low weight and high conductivity.
Graphite diamond, also known as C60, is a highly optimized version of graphite that forms strong covalent bonds through a process called intercalation. C60 is characterized by having a diameter of about 1.4 nanometers, which makes it very small compared to other carbon materials. However, its high density and stability make it an ideal material for use in high-performance applications such as fuel cells and batteries.
C60 molecules consist of a cage-like structure made up of carbon atoms that are packed tightly together. This cage structure provides a strong interlayer between the carbon atoms, making it possible for C60 molecules to form strong covalent bonds. The strength of these bonds depends on the arrangement of the carbon atoms within the cage, with stronger bonds formed when the atoms are more closely packed together.
One of the key advantages of C60 over other carbon materials is its high reactivity. This means that C60 molecules are more likely to undergo chemical reactions than other carbon materials. For example, C60 has been shown to participate in several processes related to the formation of hydrocarbons, including the breakdown of fossil fuels.
In addition to its high reactivity, C60 also has several other unique properties. For example, it exhibits excellent thermal conductivity and is able to store a large amount of energy due to its high surface area. This makes it an attractive material for use in energy storage systems such as batteries.
(What Has Stronger Carbon Carbon Bonds Graphite Diamond C60)
Overall, the properties of carbon-carbon bonds, specifically C60, make them an exciting area of research in materials science. Their ability to form strong covalent bonds through intercalation and their high reactivity make them well-suited for a variety of applications in science and technology. As our understanding of carbon-carbon bonding continues to grow, we may see even more innovative uses for this material in the future. hot tags: graphite,graphite powder,nano graphite
(What Has Stronger Carbon Carbon Bonds Graphite Diamond C60)